Abstract
Background While venetoclax (V) plus obinutuzumab (O) is a highly effective frontline CLL regimen, outcomes remain suboptimal for high-risk patients (pts). The efficacy of ibrutinib plus VO in such pts is encouraging, but cardiac and infectious toxicities (tox) are common. We hypothesized that a time-limited triplet with the more selective BTKi acalabrutinib (A) would be active and well-tolerated. We previously published results on an initial cohort unrestricted by genetic risk (Davids et al., 2021), and now report on a new cohort with enrollment restricted to high-risk pts, as well as longer follow-up on the initial cohort.
Methods Treatment (tx)-naïve pts with CLL with any genetic risk profile were initially enrolled in this investigator-sponsored, phase 2 study (cohort 1), followed by a multi-center expansion restricted to previously untreated pts with TP53-aberrant CLL (cohort 2). A starts at 100 mg BID for 28 days, followed by 2 cycles of AO (O at standard dosing), and V starts on C4D1 at 20 mg, then 50 mg on C4D2, with weekly ramp-up thereafter to 400 mg QD (total 4 week V ramp-up). AVO continues C5-7 (6 total cycles of O), and AV continues C8-15. If the primary endpoint of undetectable bone marrow MRD (BM-uMRD) CR is reached at C16D1, pts can discontinue therapy (tx); all others continue AV through C24, with the option to discontinue if BM-uMRD at C25D1. Assessments: efficacy by 2018 iwCLL criteria, toxicities (tox) by CTCAE v5, MRD by central 8-color flow (10-4 sensitivity) and NGS clonoSEQ (up to 10-6 sensitivity, Adaptive). BH3 profiling work is ongoing, performed as previously described (Ryan J et al., 2016) on peripheral blood (PB) samples after 1 and 3 cycles. Association of BCL-2 dependence with clinical response was compared by an unpaired t-test.
Results As of 13 July 2022, 68 pts were enrolled (cohort 1 all-comers: n=37, cohort 2 TP53-aberrant: n=31) in this ongoing study (NCT03580928), with median follow-up of 35 mos (range 2-45). In all 68 pts, median age 63 yrs (range 36-80); 25% ≥70 yrs; 66% male; 60% TP53-aberrant (del(17p) or mut); 24% complex karyotype (≥3 abnormalities); 74% unmutated IGHV.
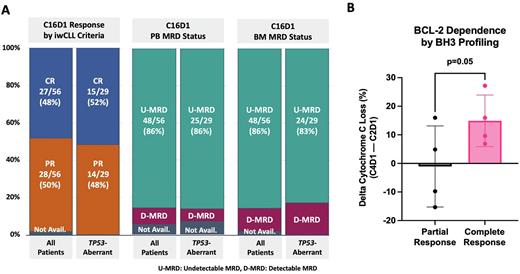
Of the 56 pts evaluable to date at C16D1, 43% (24/56) achieved the primary endpoint BM-uMRD CR. The best ORR is 98% (48% CR, 50% PR). At C16D1, MRD by flow: 86% PB-uMRD, 86% BM-uMRD (Fig 1A). In a subset of 49 pts with available samples, 59% were PB-uMRD by NGS at 10-5 sensitivity (at 10-6, 59% of pts had indeterminate results due to low cell counts). In the 29 pts with TP53-aberrant disease evaluable at C16D1, the best ORR is 100% (52% CR, 48% PR), and 45% (13/29) achieved BM-uMRD CR, MRD by flow: 86% PB-uMRD, 83% BM-uMRD (Fig 1A). There were no differences in response or MRD based on IGHV status. 79% (19/24) of pts in BM-uMRD CR electively discontinued tx after 15 cycles; median time off tx for these pts is 22.9 mos (range 1-30). 2 of the 19 pts who discontinued tx have had MRD only recurrence, and 1 had CLL disease progression; all 3 restarted tx with AV and achieved PR. 3 pts developed Richter's syndrome: 1 with DLBCL after 15 mos on study, and 2 with Hodgkin transformation (one 13 mos after completing study tx and one 12 mos into study tx; both achieved CR to Hodgkin-directed tx). 93% of all pts (63/68) remain progression-free.
In all 68 pts, all-grade heme tox included: neutropenia (75%; 37% Gr3/4), thrombocytopenia (73%; 28% Gr3/4), anemia (49%, 3% Gr3). Non-heme tox: headache (78%, 1% Gr3), fatigue (76%, 1% Gr3), bruising (66%, all Gr1/2), nausea (49%, all Gr1/2), diarrhea (40%, 4% Gr3), infusion-related reaction (30%, 4% Gr3), hypertension (27%; 9% Gr3), increased ALT (27%, 1% Gr3), arthralgia (25%, all Gr1), and infection (6% Gr3; 1 case of Gr5 COVID-19 pneumonia). 2.9% (2/68) pts had afib. 14 pts (21%) required dose-reduction of either only A (4%), only V (9%), or both drugs (7%).
BH3 profiling is ongoing, and initial data suggest that pts who later went on to achieve CR may have a greater increase in BCL-2 dependence at C4D1 than pts who achieved PR as best response (p=0.05, Fig 1B).
Conclusion AVO is a highly active, well-tolerated triplet in a frontline CLL population enriched for high-risk disease, with 83% of TP53-aberrant pts achieving BM-uMRD after 15 mos of tx. Responses are durable, with 93% PFS in all pts at a median follow-up of nearly 3 yrs. Low rates of cardiac and infectious tox were observed. Our data support continued investigation of the MRD-guided, time-limited AVO triplet.
Disclosures
Crombie:Incyte: Consultancy; Kite: Consultancy; Bayer: Research Funding; Abbvie: Research Funding; Roche: Research Funding; Merck: Research Funding; Karyopharm: Consultancy. Montegaard:Pharmacyclics: Consultancy; Abbvie: Consultancy; AstraZeneca: Consultancy. Jacobson:Kite/Gilead: Consultancy, Research Funding; Novartis: Consultancy; BMS/Celgene: Consultancy; Bluebird Bio: Consultancy; Epizyme: Consultancy; Ipsen: Consultancy; Instil Bio: Consultancy; ImmPACT Bio: Consultancy; Caribou Bio: Consultancy; Morphosys: Consultancy; Miltenyi: Consultancy; Abintus Bio: Consultancy; Pfizer: Research Funding. LaCasce:Research to Practice: Consultancy; Seattle Genetics: Consultancy. Armand:Infinity: Consultancy; Pfizer: Consultancy; Adaptive: Consultancy, Research Funding; Affimed: Consultancy, Research Funding; BMS: Consultancy, Honoraria, Research Funding; Merck: Consultancy, Honoraria, Research Funding; ADC Therapeutics: Consultancy; Celgene: Consultancy; Morphosys: Consultancy; Daiichi Sankyo: Consultancy; Miltenyi: Consultancy; Tessa: Consultancy; Genmab: Consultancy; C4: Consultancy; Enterome: Consultancy; Regeneron: Consultancy; Epizyme: Consultancy; AstraZeneca: Consultancy; Genentech: Consultancy, Research Funding; Roche: Research Funding; Xencor: Consultancy; Tensha: Research Funding; Otsuka: Research Funding; Sigma Tau: Research Funding; IGM: Research Funding; Kite: Research Funding. Arnason:Bristol Myers Squibb: Speakers Bureau. Brown:Abbvie, Acerta/Astra-Zeneca, BeiGene, Bristol-Myers Squibb/Juno/Celgene, Catapult, Eli Lilly, Genentech/Roche, Hutchmed, iOnctura, Janssen, MEI Pharma, Pharmacyclics: Consultancy; BeiGene, Gilead, Loxo/Lilly, MEI Pharma, SecuraBio, Sun, TG Therapeutics: Research Funding. Davids:BeiGene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Adaptive Biotechnologies: Consultancy, Membership on an entity's Board of Directors or advisory committees; Eli Lilly and Company: Consultancy, Membership on an entity's Board of Directors or advisory committees; Merck: Consultancy; Ascentage Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel expenses, Research Funding; Ono Pharmaceuticals: Consultancy; Novartis: Research Funding; Takeda: Consultancy; Bristol-Myers Squibb: Consultancy, Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Research to Practice: Honoraria; Verastem: Consultancy, Research Funding; TG Therapeutics: Consultancy, Research Funding.
OffLabel Disclosure:
Acalabrutinib, venetoclax, and obinutuzumab were investigated in combination.
Author notes
Asterisk with author names denotes non-ASH members.